We play with natural polymers to design nano-size metamaterials capable
of selectively trapping specific biomolecules
 bioMIP
bioMIP
Sustainable, selective, biocompatible nanotraps engineered from natural biomaterials
We play with natural polymers to design nano-size metamaterials capable
of selectively trapping specific biomolecules
BioMIPs are biopolymer-based molecularly imprinted nanoparticles capable of selectively capturing biomarkers, proteins, or small molecules.
They combine the precision of synthetic receptors with the biocompatibility and sustainability of natural materials such as silk fibroin and gelatin.
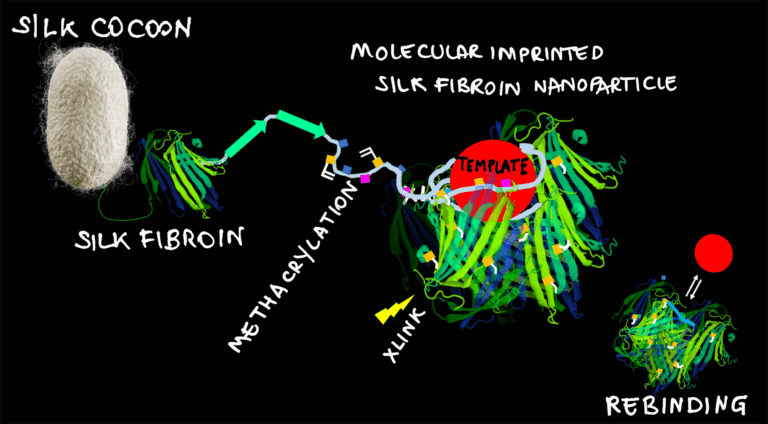
This work introduces molecularly imprinted nanoparticles made entirely from natural silk fibroin, offering a fully biocompatible alternative to traditional polymer-based MIPs. By using template-assisted imprinting, each nanoparticle forms a single, selective, high-affinity recognition site (Kd ≈ 57 nM) towards Albumin.
Controlled synthesis conditions—defined through response surface modeling—enabled precise size tuning (≈50 nm and ≈100 nm). The resulting silk nanoMIPs were confirmed to be nontoxic and could be integrated onto silk microfibers and nanofibers, demonstrating a versatile route for adding specific molecular recognition features to natural-material platforms.
A sustainable, safe, and versatile alternative to synthetic plastic antibodies.
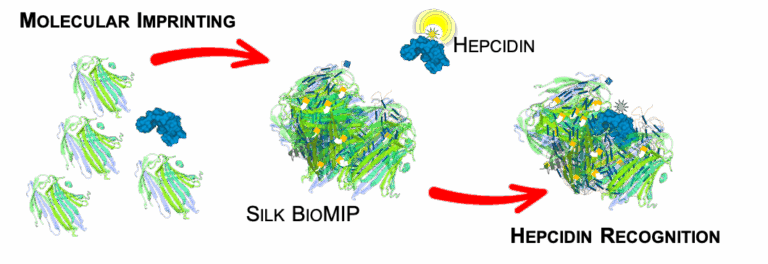
Building on earlier work with methacrylated silk fibroin, this study demonstrates the imprinting of silk-based bioMIPs using hepcidin, a key regulator of iron metabolism, as the template.
A uniform nanoparticle population (~50 nm, PDI < 0.2) was obtained, showing selective binding to hepcidin with a dissociation constant of Kd ≈ 3.6 × 10⁻⁷ M and an average of two binding sites per bioMIP.
When applied in a competitive assay in serum, these bioMIPs enabled reliable hepcidin quantification with a detection range of 1.01 × 10⁻⁷ to 6.82 × 10⁻⁷ M and a limit of detection of 3.29 × 10⁻⁸ M.
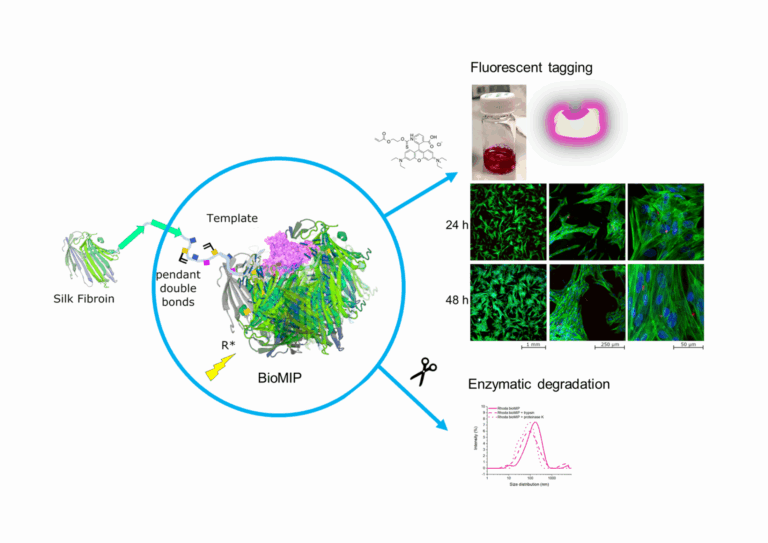
Building on the initial demonstration that silk-fibroin bioMIPs can selectively recognize human serum albumin, this study extends that foundation with two key advancements. First, it introduces a robust method for post-synthetic fluorescent tagging, allowing bioMIPs to be directly visualized within cells and tissues. Second, it investigates their enzymatic degradation, confirming that these nanoparticles can be naturally broken down into safe byproducts
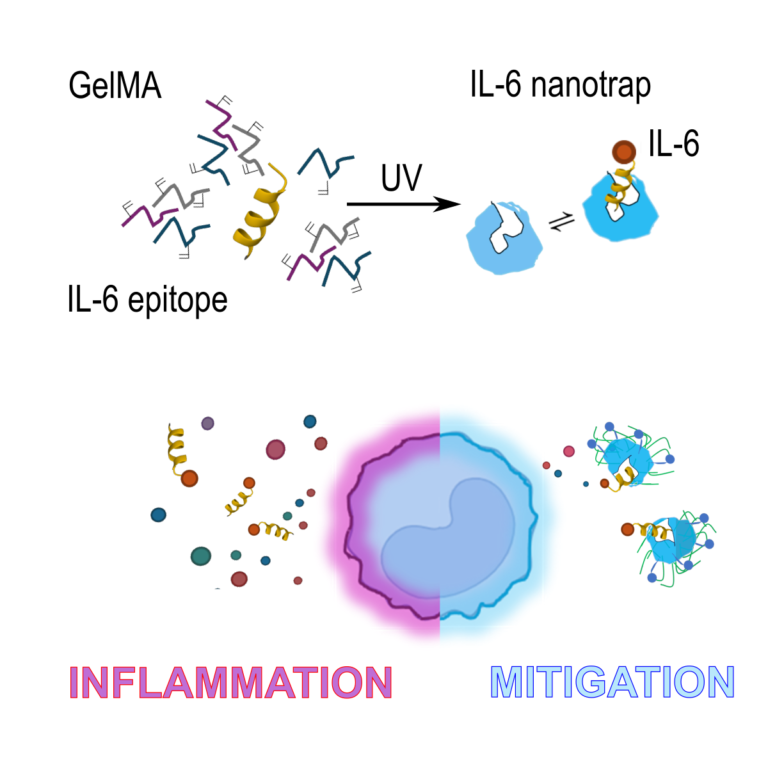
Gelatin methacryloyl (GelMA) was harnessed to create nanotraps ve specically designed to scavenge interleukin-6 (IL-6) in infammation models. This innovative approach leverages safe and biocompatible protein-based biomaterials, paving the way for a novel class of therapeutic solutions with signi cant clinical potential.
This review charts the evolution of molecularly imprinted polymers made from natural biopolymers, drawing on research from the early 1980s to today. It examines major biopolymer families—glucans, chitosan, alginates, proteins, and nucleic acids—highlighting how each material can be engineered, imprinted, and applied.
By assessing synthetic strategies, performance, and limitations, the review provides a critical framework for understanding how biopolymers can deliver highly selective, biocompatible, and sustainable recognition systems. Together, these insights position biopolymer-based MIPs as a powerful and expanding frontier in advanced molecular imprinting.


The bioMIP’s technology has been financed from the following projects: